| Product: | Plerixafor | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Catalog Number: | 16854 | ||||||||
| CAS Number: | 110078-46-1 | ||||||||
| Synonyms: | 1,1’-[1,4-Phenylenebis(methylene)]bis-1,4,8,11-tetraazacyclotetradecane | ||||||||
| Pricing: |
|
||||||||
| Formula: | C28H54N8 | ||||||||
| Chemical Purity: | 98% | ||||||||
| Molecular Weight: | 502.78 | ||||||||
| Structure: |
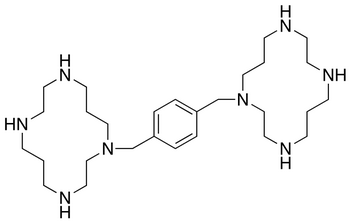
|
||||||||
| Appearance: | White powder | ||||||||
| Category: | Various Intermediates | ||||||||
| Stability: | Stable under recommended storage conditions. | ||||||||
| Storage: |
Refrigerator |
||||||||
| Transportation: |
Non-hazardous for transport |
||||||||
| Literature References: |
DiPersio, J.F., Stadtmauer, E.A., Nademanee, A., Micallef, I.N.M., Stiff, P.J., Kaufman, J.L., Maziarz, R.T., Hosing, C., Fruehauf, S., Horwitz, M., Cooper, D., Bridger, G., Calandra, G., Plerixafor and G-CSF versus placebo and G-CSF to mobilize hematopoietic stem cells for autologous stem cell transplantation in patients with multiple myeloma. Blood, 2009, 113(23):5720-5726.; Dugan, M.J., Maziarz, R.T., Bensinger, W.I., Nademanee, A., Liesveld, J., Badel, K., Dehner, C., Gibney, C., Bridger, G., Calandra, G., Safety and preliminary efficacy of plerixafor (Mozobil) in combination with chemotherapy and G-CSF: an open-label, multicenter, exploratory trial in patients with multiple myeloma and non-Hodgkin's lymphoma undergoing stem cell mobilization. Bone Marrow Transplantation, 2010, 45:39-47. |
||||||||
| MSDS: | |||||||||
| Applications: | An immunostimulant, used to mobilize hematopoietic stem cells in cancer patients. |
