| Product: | Eflornithine HCl hydrate | |
|---|---|---|
| Catalog Number: | 17462 | |
| CAS Number: | 70052-12-9 (free base) | |
| Synonyms: |
DL-α-Difluoromethylornithine hydrochloride hydrate; DMFO hydrochloride hydrate; 2-(Difluoromethyl)ornithine hydrochloride hydrate |
|
| Pricing: |
|
|
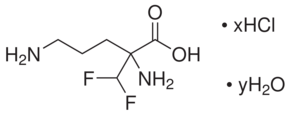
| Formula: | C6H12F2N2O2 . xHCl . yH2O | |
| Chemical Purity: | >97% | |
| Molecular Weight: | 182.17 | |
| Structure: |
|
|
| Appearance: | White solid | |
| Category: | Anti-cancer compounds | |
| Stability: | Stable under recommended storage conditions | |
| Storage: |
Keep container tightly closed in a dry and well-ventilated place. |
|
| Transportation: |
Non-hazardous for transport |
|
| Literature References: |
Sharma, S., et al., , A quantitative angiogenesis model for efficacy testing of chemopreventive agents. Anticancer Res. 21, 3829-3837, (2001); Takahashi, Y., et al., α-Difluoromethylornithine induces apoptosis as well as anti-angiogenesis in the inhibition of tumor growth and metastasis in a human gastric cancer model. Int. J. Cancer 85, 243-247, (2000); Jasnis, M.A., et al., Polyamines prevent DFMO-mediated inhibition of angiogenesis. Cancer Lett. 79, 39-43, (1994); Takigawa, M., et al., Tumor angiogenesis and polyamines: α-difluoromethylornithine, an irreversible inhibitor of ornithine decarboxylase, inhibits B16 melanoma-induced angiogenesis in ovo and the proliferation of vascular endothelial cells in vitro. Cancer Res. 50, 4131-4138, (1990); Merck 14,3522 |
|
| MSDS: | ||
| Description: |
Difluoromethylornithine (Eflornithine) inhibits polyamine biosynthesis by the selective, irreversible inhibition of ornithine decarboxylase (ODC). A chemoprotective agent that blocks angiogenesis. |
